Overcoming optimisation hurdles of high-throughput FMT assay
Many scientists face hurdles optimising their FMT assay, see how Newcells has solved those.
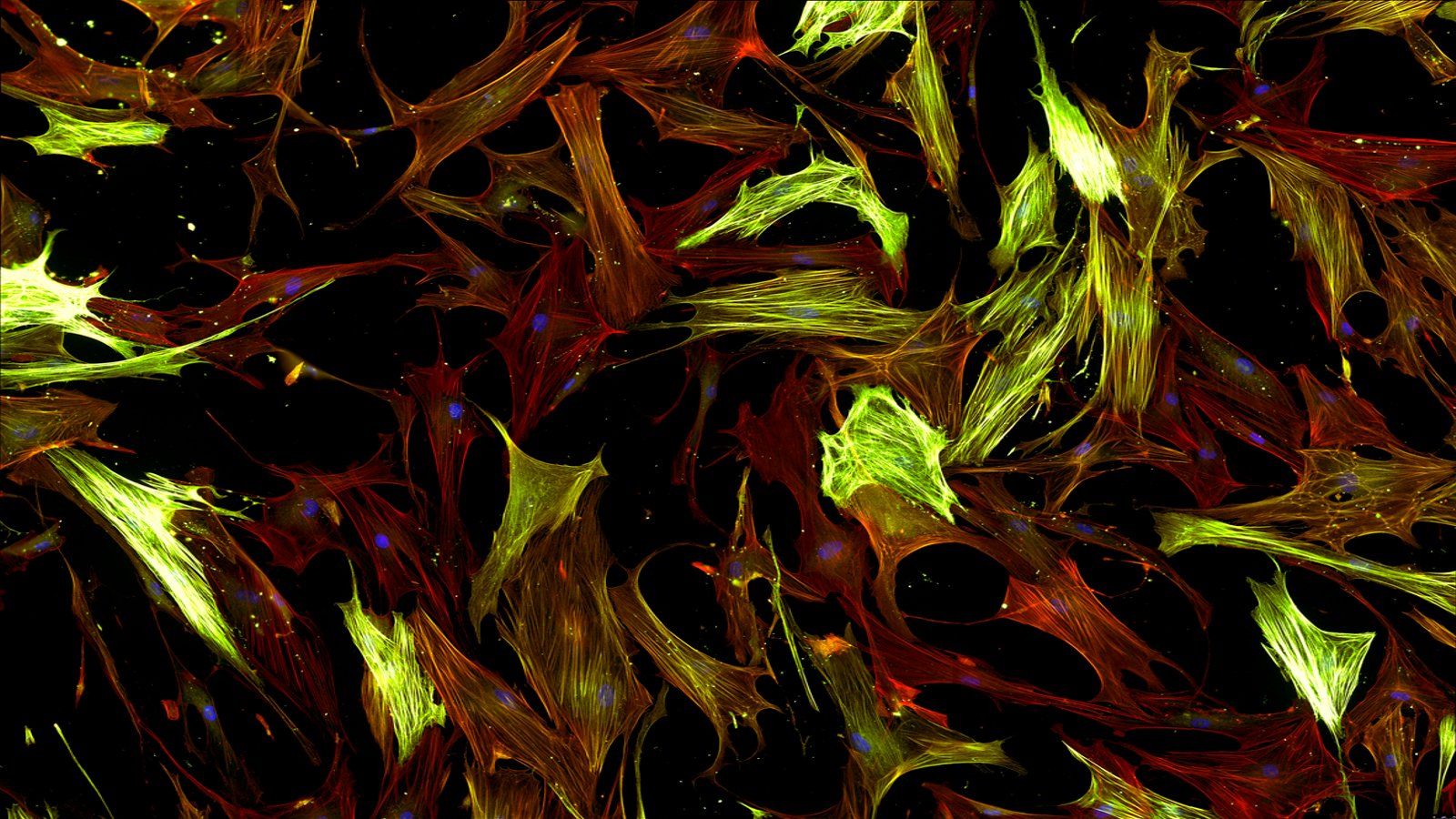
Several hurdles faced by many scientists, when developing assays and models for drug discovery, were highlighted at the 2023 European Respiratory Society (ERS) Congress. Understanding and investigating disease processes such as fibroblast-to-myofibroblast (FMT) transition in idiopathic pulmonary fibrosis (IPF), is key to enabling effective compound screening. Whilst the authors successfully developed a high-throughput FMT assay for compound screening, they reported significant difficulties in the process.
Interestingly, the head of our lung platform, Dr. Megan Webster presented how we addressed these familiar and recurring challenges at the recent antifibrotic drug development (AFDD) conference in Boston, USA. Our team introduced mechanical washing for 384-well plates, a prevalent methodology, which led to significant cell detachment and increased assay variability. We overcame these recurrent issues via optimisation of plate coatings, cell number, and media composition as well as the inclusion of a macromolecular crowding (MMC) agent. See our poster for further details.
Further optimisation of immunocytochemistry protocols, high-content imaging parameters, and segmental analysis masks was also performed to develop a robust, reproducible, high-throughput FMT assay. To save valuable time and resources, why not try our fully standardised FMT assay service.
Contact us to outsource your FMT-assay studies to Newcells Biotech today!
Share on social media:
Don't miss out on our latest innovations: follow us on Linkedin
Dr Megan Webster
7th December, 2023
Lung



