Kidney proximal tubule cell model for safety/efficacy and transporter studies
Newcells Biotech offers a comprehensive platform for in vitro renal studies from transporter studies, DDI to nephrotoxicity.
Overview
A unique primary proximal tubule cell model derived from fresh kidney tissue with high renal transporter expression levels.
Characterization
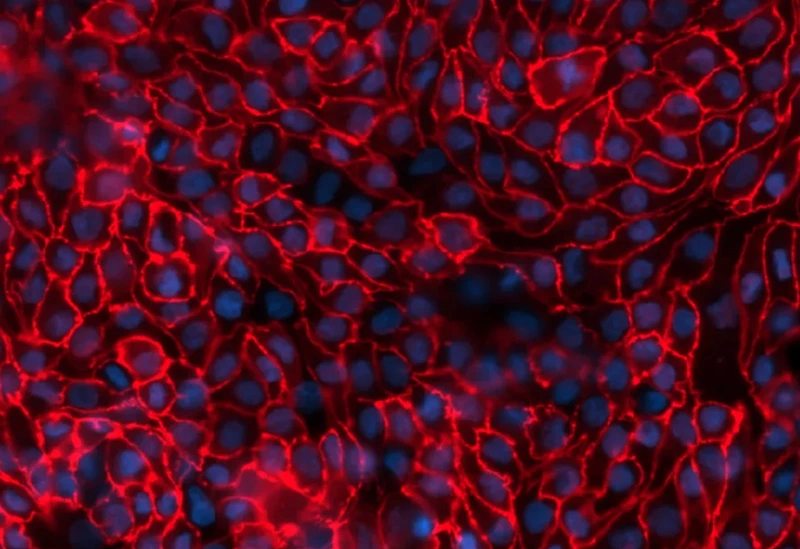
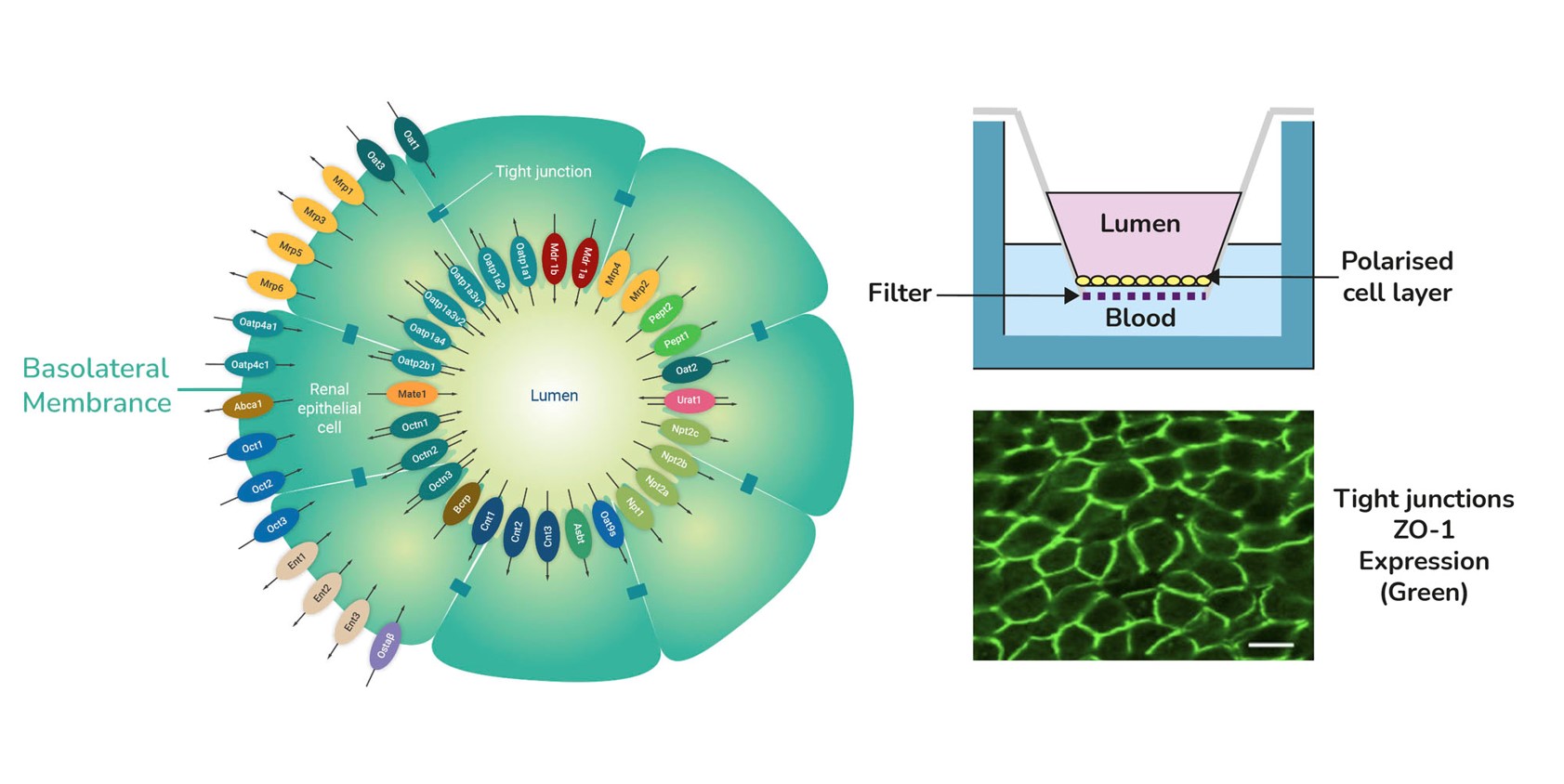
aProximate™ PTCs are primary cells derived from fresh human kidney tissue. They are grown on Transwells® and express the functional properties of a polarised cell layer with tight junctions.
In contrast to other models, aProximate™ PTCs retain high expression of the key renal transporters involved in drug handling including Megalin and Cubilin. Studies with aProximate™ give you a detailed mechanistic understanding of how drugs are transported and eliminated through the kidney. The model can be used to carryout drug-drug interaction studies to inform clinical trial design.
Key Features
- Functional properties of polarised cell layer with tight junctions
- Expressed Megalin and Cubilin
- Available in multiple species (rat, dog, monkey, human)
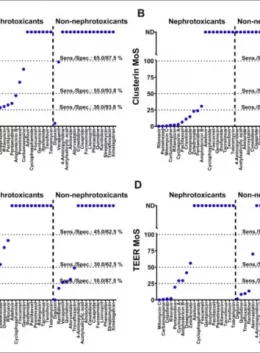
- Fast and reliable Nephrotoxicity service
Available Analytical Readouts
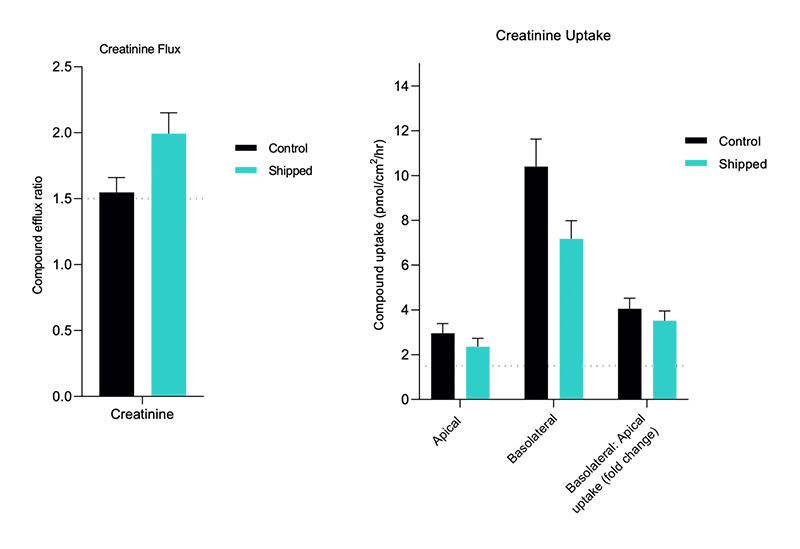
- Flux and net transport measurements
- Measurement of intracellular drug and metabolite concentrations
- Identification of transporter-mediated drug-drug interactions
- Early renal damage biomarkers
- Cell viability