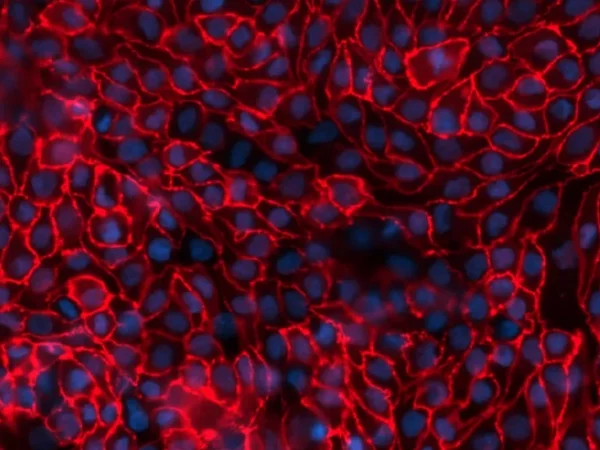
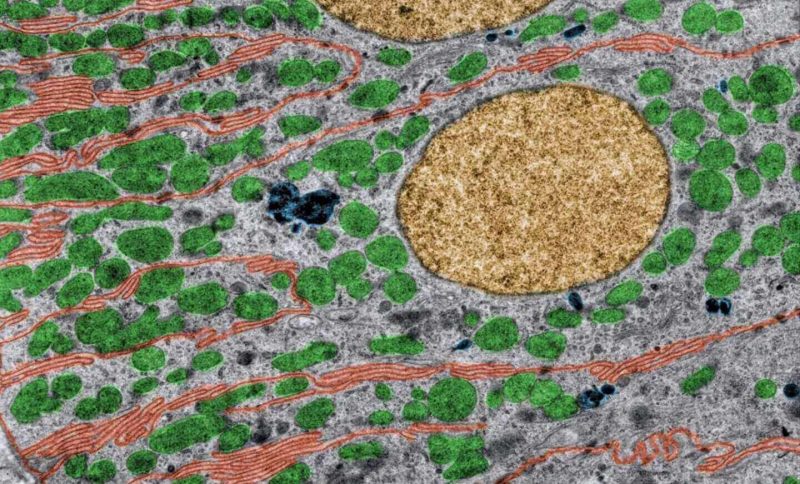
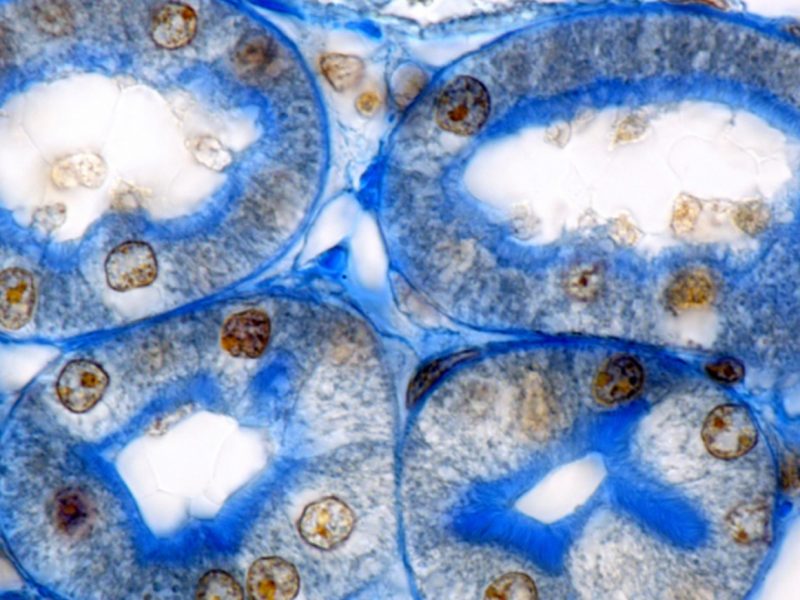
Cross-species comparison of drug handling with near-physiological kidney transporter model
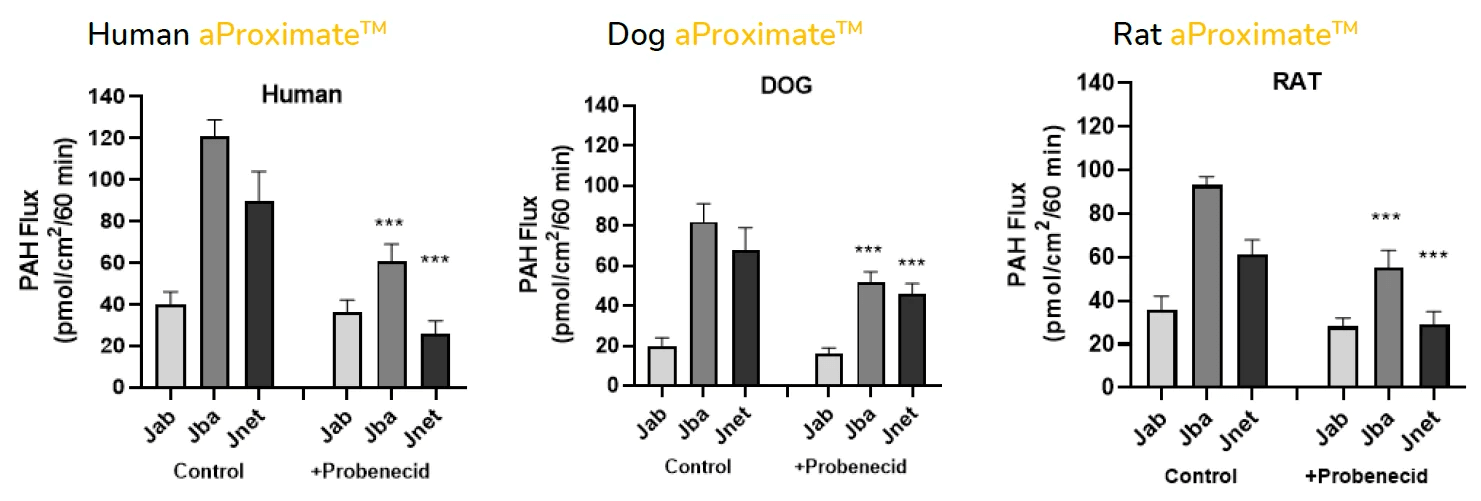
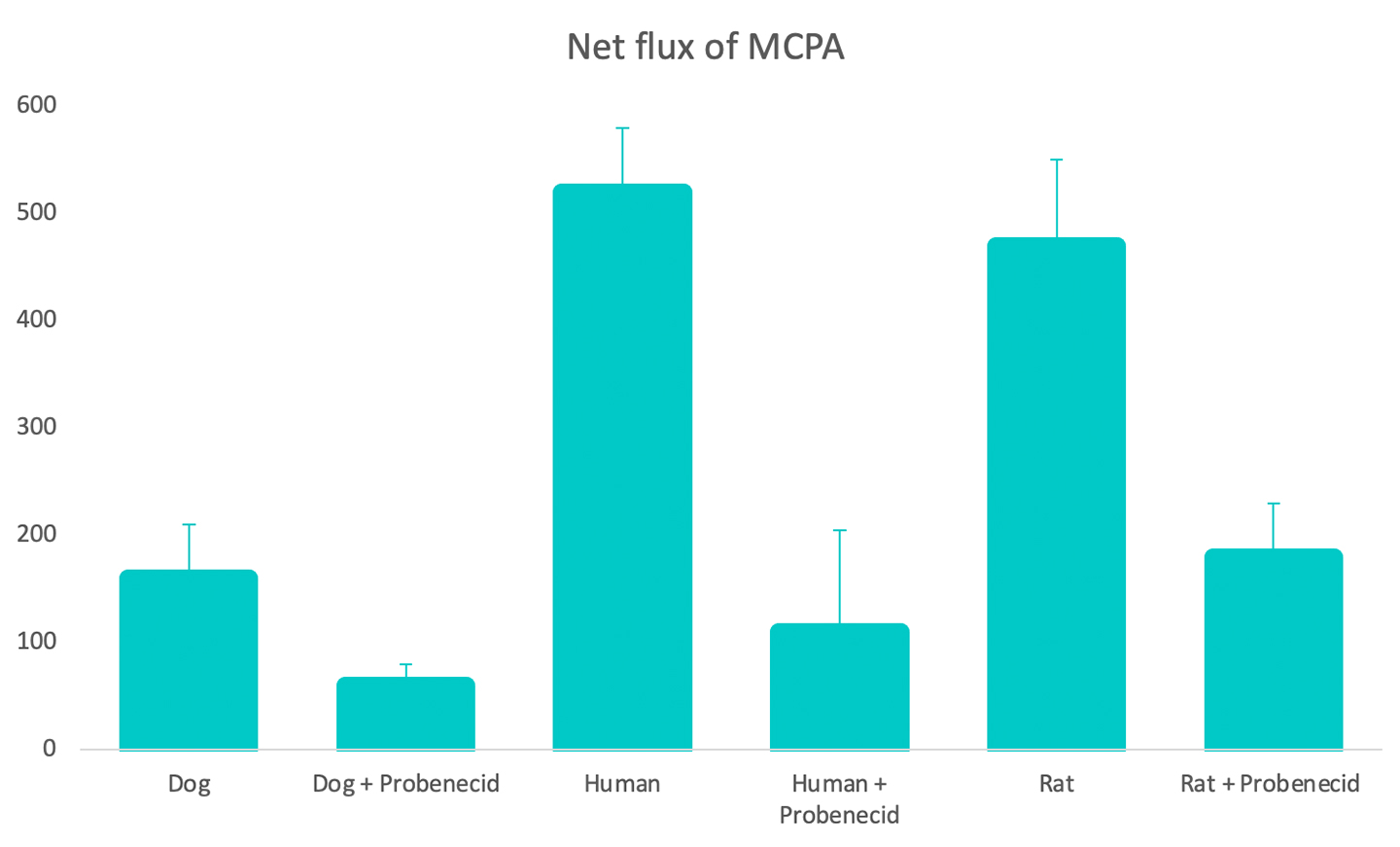
New drug molecules are usually tested in vivo in two preclinical species to gain a more detailed understanding of how they are transported and eliminated through the kidney and also to mitigate risk of renal toxicity in humans. However, these animal models are not always predictive of human responses. To address this problem, robust, comparative in vitro kidney transporter models, such as aProximate™, are powerful tools to compare different species and are much needed to improve in vitro-in vivo extrapolation (IVIVE).
Service outputs
Species differences in:
- Apical to Basal (Jab) and Basal to Apical (Jba) flux
- Net transport
- Renal toxicity
Don't miss out on our latest innovations: follow us on Linkedin