In vitro evaluation of ocular toxicity and phenotypic rescue
Tissue
Retina ModelsThe problem and data requirement
Our client wished to evaluate the retinal toxicity of a pair of lead compounds and understand how each affects the retina.
Solution
Step 1:
Our experts worked with the customer to understand the problem
The client needed a retina model to rapidly evaluate the retinal toxicity of two molecules and understand possible differences. We proposed to determine how each compound affects cell viability as well as the cellular composition of our iPSC-derived retinal organoid model.
Step 2:
We designed a study protocol and design that efficiently provided data of value
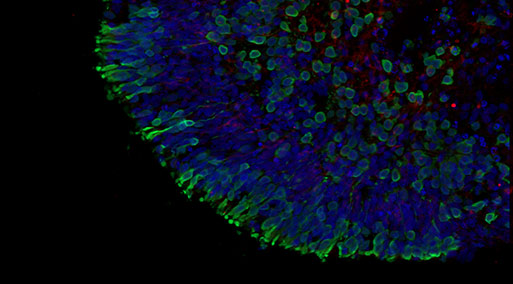
The experimental plan involved testing compound induced toxicity using mature healthy retinal organoids differentiated for 150 days exposing the organoids for 72 hours. The readouts included retinal organoid viability assays and immunofluorescence with 6 key retinal markers to assess the organoid cells composition (ganglion cells, photoreceptors, amacrine cells, horizontal cells and Muller glia).
Step 3:
From initiation to data in less than two months
The study includes the experimental phase, data processing and analysis as well as a data summary presentation.
DATASET: 4 compounds, 1 positive control, 100 retinal organoids, 7 immunofluorescence staining per compound, ATP depletion and LDH release for each compound
The study was completed in just under 2 months.
Step 4:
We deliver a detailed report with reliable dataset
We delivered a robust dataset which contained a large set of immunofluorescence images
Outcomes for the client
Our analysis confirmed retinal toxicity of one of the compounds and we provided insights to the client as to how the compound affects different cell types.