Pulmonary toxicity – a challenge for drug development
Despite the common assumption that adverse drug reactions usually affect the kidney and liver, over 600 medications have been reported to induce lung toxicity.1 Due to the large surface area, lungs are particularly susceptible to toxicity. Furthermore, the mechanisms by which they metabolise certain substances2, such as antibiotics, anti-inflammatories, anticancer drugs and drugs targeting the cardiovascular system also lead to detrimental effects, resulting in 4-10% of patients developing pulmonary toxicity.3
“There are more than 600 medications known to induce pulmonary toxicity” (1)
Due to the rapid increase of new chemicals released into the market (approximately 3,000 annually)4, the Registration, Evaluation, Authorization and restriction of Chemicals (REACH) reported that 90,000 vertebrate animals are employed annually in toxicity testing in Europe alone.5 While most toxicology studies are performed by exposing animals to high-dose concentrations and tracking toxic responses to substances, these studies often don’t effectively predict reactions in humans.2,6 This is either due to interspecies metabolic and genetic differences or an inability to test multiple drug-to-drug interactions in an animal model due to cost and reduction of animal testing principles. 6 These disparities increase the possibility of adverse reactions in humans and this is why environmental and toxicological organisations are seeking predictive human cell models to improve their drug screening process for pulmonary disease with novel in vitro approaches to toxicity screening.2,6
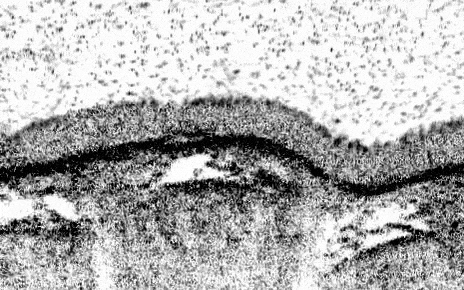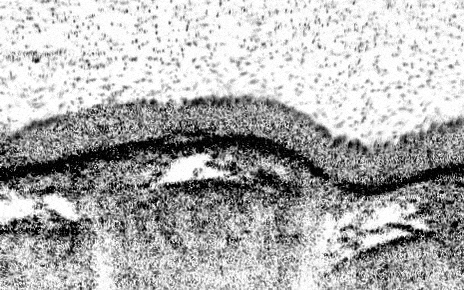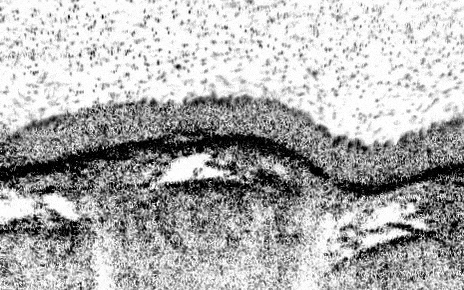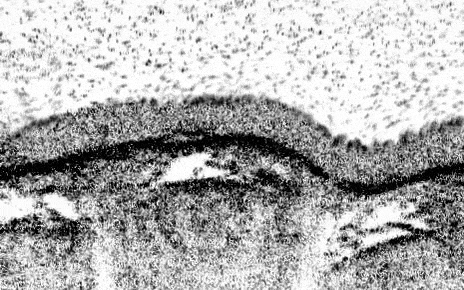
Newcells Biotech R& D program has developed a functional primary lung 3–D model of the small airways, containing all of the key epithelial cell types. The structure of our Small Airway Epithelial Cell (SAEC) in vitro model mimics the in vivo airways of the lung by presenting tight epithelial junctions and a structured pseudostratified epithelium of functional basal cells, goblet cells, club cells and beating ciliated cells with a mucus layer on the apical side visible by brightfield microscopy. This air-liquid interface, lung airway model overcomes the drawbacks of previous in vitro models, which contain less cell types and had reduced functionality.
Newcells’ lung 3-D in vitro model has been designed to increase the available screening capacity to aid industry in more extensively testing for pulmonary toxicity. Our SAEC model provides a powerful in vitro tool to model airway physiology and respiratory diseases, assess drug toxicity, determine therapeutic mechanisms of action, and confirm target engagement. Cultured on 24-well permeable support membranes, we are currently offering readouts including TEER, cytokine release, gene expression modifications, immunocytochemistry, cell viability, and compound toxicity. We are currently working on optimising the model in a 96-well format.
References:
- Drug-Induced Pulmonary Toxicity: Practice Essentials, Background, Pathophysiology [Internet]. [cited 2020 Feb 6]. Available from: https://emedicine.medscape.com/article/1343451-overview
- Schwaiblmair M, Behr W, Haeckel T, Märkl B, Foerg W, Berghaus T. Drug Induced Interstitial Lung Disease. Vol. 6, The Open Respiratory Medicine Journal. 2012.
- Ellis SJ, Cleverley JR, Müller NL. Drug-Induced Lung Disease. Am J Roentgenol [Internet]. 2000 Oct [cited 2020 Feb 6];175(4):1019–24. Available from: http://www.ajronline.org/doi/10.2214/ajr.175.4.1751019
- Bengtsson BMI. Global Trends – Threats or Challenges to Future Food Security and Food Safety. In: Agricultural Research at the Crossroads [Internet]. CRC Press; 2018 [cited 2020 Feb 7]. p. 97. Available from: https://books.google.co.uk/books?id=gJWwDwAAQBAJ&printsec=frontcover#v=onepage&q&f=false
- Rovida C, Hartung T. Re-evaluation of animal numbers and costs for in vivo tests to accomplish REACH legislation requirements for chemicals – A report by the transatlantic think tank for toxicology (t4). ALTEX. 2009;26(3):187–208.
- Krewski D, Acosta D, Andersen M, Anderson H, Bailar JC, Boekelheide K, et al. Toxicity testing in the 21st century: A vision and a strategy. Vol. 13, Journal of Toxicology and Environmental Health – Part B: Critical Reviews. 2010. p. 51–138.
Talk to our leading lung experts
Book a scientific discussionShare on social media:
Don't miss out on our latest innovations: follow us on Linkedin



